Abstract
Epstein Barr Virus (EBV) is a gammaherpesvirus responsible for a broad spectrum of human diseases, including hematologic malignancies and autoimmune disorders. EBV maintains lifelong infection by entering an endosomal state of latency inside its host cell. In response to external stimuli EBV undergoes lytic activation, resulting in vast gene expression changes, DNA replication, and eventually release of infectious viral particles. DNA methylation of the EBV genome is essential to regulating its life cycle. Both lytic and latent life stages are key determinants of EBV-driven disease, but current clinical assays for EBV are limited to detecting the presence of EBV and do not provide important information on EBV lytic state. Thus, making clinical decisions based on EBV detection alone has remained challenging.
To address these limitations, we have developed an EBV analysis pipeline utilizing nanopore sequencing from amplification-free, native DNA that directly interrogates CpG dinucleotide DNA methylation on the EBV genome. Our workflow generates highly accurate EBV methylation calls for the majority of CpGs across the EBV genome. We also deployed a quantitative mass-spectrometry-based methylation assay (EpiTYPER) to validate our data and provide broader analysis of a representative subset of methylation sites (n=123 methylation sites from n=25 EBV loci).
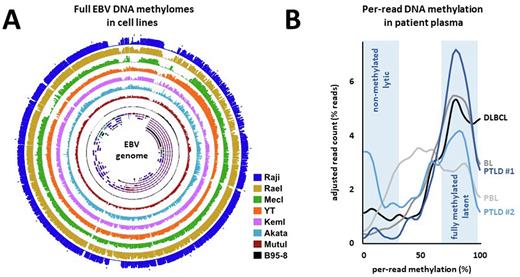
To initially investigate the relationship between locus-specific EBV DNA methylation and EBV gene expression, we compared DNA methylation and RNA sequencing profiles in EBV-positive cell lines (n=7). We correlated global EBV DNA methylation levels and the overall patterns of latent versus lytic EBV gene expression. We found that average methylation levels are related to the ratio of latent versus lytic gene expression, establishing nanopore-derived DNA methylation as a method to quantify latent and lytic states in a given sample.
We next shifted our approach to bin nanopore reads based on the average methylation of all CpGs per read, hypothesizing that latent/lytic viral states derive from distinct subpopulations of EBV endosomes that contain methylated versus unmethylated DNA. We uncovered widespread co-occurrence of high and low methylated viral DNA in cell lines, indicating that EBV methylation states exist in defined subfractions with either present or absent DNA methylation. In cell models of latent EBV infection (Akata, Kem, MEC1, Mutu, Rael) this approach showed that methylation states dynamically responded to lytic induction and selective pressure via antiviral treatment with ganciclovir. Of note, we observed a shift in methylation without the appearance of partially methylated DNA, indicating that EBV lytic activation/replication is associated with the appearance of unmethylated EBV DNA, and not gradual demethylation of endosomes in the latent viral state.
Finally, we aimed to obtain measurements of overall EBV status in patients, using nanopore sequencing of cell-free DNA (cfDNA) obtained from patient plasma. We analyzed patients diagnosed with different EBV-positive diseases from The Ohio State University Medical Center, including n=3 patients with lymphomas (Burkitt, plasmablastic and diffuse large B cell lymphoma) and n=2 patients with post-transplant lymphoproliferative disorder (PTLD). The patient samples with lymphoma displayed a near uniformly high methylation profile, with little evidence of unmethylated DNA/lytic replication. One of the PTLD patients analyzed showed a distinct bi-modal distribution of per read EBV methylation values, indicating the presence of lytic replication in a significant proportion of cells.
In conclusion, nanopore sequencing analysis of methylated versus unmethylated EBV DNA subfractions is a quantitative method for determining viral states. In patient samples, nanopore sequencing revealed methylation heterogeneity in cfDNA and may be a useful advancement compared to standard clinical EBV detection. Quantifying the relative ratio of latent versus lytic virus in a liquid biopsy sample could be used to guide therapy selection for patients displaying EBV-positivity.
Disclosures
Baiocchi:Viracta Therapeutics: Consultancy, Current holder of stock options in a privately-held company, Membership on an entity's Board of Directors or advisory committees; eLife (Journal): Other: Editorial board; CODIAK Biosciences: Research Funding; Atara Biotherapeutics: Consultancy, Membership on an entity's Board of Directors or advisory committees. Blachly:Astellas: Consultancy, Membership on an entity's Board of Directors or advisory committees; MingSight Pharmaceuticals: Research Funding; AbbVie: Consultancy, Membership on an entity's Board of Directors or advisory committees; KITE Pharma: Consultancy, Membership on an entity's Board of Directors or advisory committees; AstraZeneca: Consultancy, Membership on an entity's Board of Directors or advisory committees; INNATE Pharma: Consultancy, Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal